|
The electron has such a small mass that we do not consider the mass of electrons in the atomic mass, the electron has a mass that is 0.00054 compared to the mass of protons and neutrons of 1. An oxygen has 8 protons with 8 positive charges and 8 negatively charged electrons orbiting around it in shells. This however does not consider the mass of the 8 electrons. Shahen Akopyan 2E wrote:Yes, I believe it would be best to keep the same number of digits you see on the periodic table for molar mass in your calculation steps and worry about significant figures at the end depending on the sig figs for the values given in the original problem.This way there would be less variance with the solutions. The mass of 16 is made up of 8 protons and 8 neutrons. By looking at the mass number of 16 we can also deduce that there are 8 other particles in the nucleus, those 8 other particles are neutrons as neutrons and protons both have the same mass of 1. Using this it can be seen that Carbon has 6 protons in its nucleus and subtracting the atomic number from the atomic mass (12-6) it can be seen that there are 6 neutrons in the nucleus.įor example, Oxygen has an atomic number of 8 indicating it has 8 protons in its nucleus. Carbon has a mass listed as 12 and an atomic number of 6. The example of Carbon and chlorine illustrates this well. We can use this number to understand how many protons and neutrons there are in the nucleus. This number is always the larger of the two represented in the periodic table. Each element square contains all 118 of elements with the element number, symbol, name, atomic mass, and most common oxidation number. Reviewing the way of finding the charge on an atom may help, too. This periodic table is a periodic table with charges PDF optimized to fit on a single sheet of paper. – Visit iupac.Atomic mass refers to the mass of both the protons and neutrons. This periodic table with charges is a useful way to keep track of the most common oxidation numbers for each element. Practical applications of isotopic measurements and technologies are included for the following fields: forensic science, geochronology, Earth-system sciences, environmental science, and human health sciences, including medical diagnosis and treatment. The background color scheme of cells categorizes the 118 elements into four groups: (1) white indicates the element has no standard atomic weight, (2) blue indicates the element has only one isotope that is used to determine its standard atomic weight, which is given as a single value with an uncertainty, (3) yellow indicates the element has two or more isotopes that are used to determine its standard atomic weight, which is given as a single value with an uncertainty, and (4) pink indicates the element has a well-documented variation in its atomic weight, and the standard atomic weight is expressed as an interval.Īn element-by-element review accompanies the IPTEI and includes a chart of all known stable and radioactive isotopes for each element. Color-coded pie charts in each element cell display the stable isotopes and the relatively long-lived radioactive isotopes having characteristic terrestrial isotopic compositions that determine the standard atomic weight of each element. The IPTEI is intended to hang on the walls of chemistry laboratories and classrooms.Įach cell of the IPTEI provides the chemical name, symbol, atomic number, and standard atomic weight of an element. The IPTEI is modeled on the familiar Periodic Table of the Chemical Elements. Name chemical element: Symbol: Atomic number - Name alphabetically: 1.0079: Hydrogen: H: 1 - Atomic number: 4. Nominations Open for the 9th Polymer International-IUPAC Award Ībstract: The IUPAC Periodic Table of the Elements and Isotopes (IPTEI) was created to familiarize students, teachers, and non-professionals with the existence and importance of isotopes of the chemical elements. the periodic chart sorted by: Atomic Mass.eTOC ‘Chemistry International’ – Jan-Mar 2024. /chemical-periodic-table-of-elements-with-color-cells-vector-illustration-840464844-ff61af51081b4fb1959504105e232e01.jpg)
The 2024 IUPAC-Richter Award Goes to Craig M.IUPAC Standards Online – FREE ACCESS IN 2024. 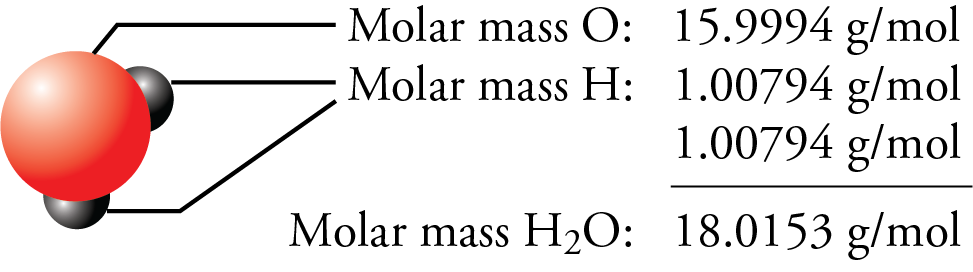
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
