|
29 In addition to provoking action potentials, electrical stimulation alters the membrane potential of neurons and other cell types exposed to electric fields, thereby altering electrochemical properties of the segments affected. This activation can be measured as action potentials propagated antidromically in peripheral nerves, 23, 24 as epidural action potentials, 25, 26 as somatosensory evoked potentials recorded on the scalp, 25 and as muscle twitches in limb and trunk muscles, 27, 28 and felt by patients as paresthesias. 14, 21, 22 Conventional SCS preferentially activates large Ap dorsal column axons. The bioelectrical properties of the spinal cord have received signficant attention, and a number of reviews have been published on this topic. Large diameter axons have low thresholds for firing action potentials, and thus are preferrentially activated over smaller fibers. As electrodes are typically placed near the physiological midline of the dorsal columns (except in the case of DRG stimulation), electrical stimulation causes activation of dorsal column axons, resulting in orthodromic and antidromic transmission of action potentials that generate segmental and supraspinal effects 2, 14, 17– 20 ( Figure 1). In the case of excitable membranes, such as those found in nearby dorsal column axons, the electric field can trigger one or more action potentials, depending on the bioelectrical properties of the axon (diameter, myelination status, and electrical threshold). The applied fields change the electrical potential across membranes based on the properties of tissues near the electrode, such as the dura, layer of cerebrospinal fluid, and white matter. Fundamentally, SCS, regardless of type, involves the generation of electric fields between metal contacts residing in the epidural space. 14– 16 Some of the new waveforms, such has high-frequency stimulation, have challenged our ability to elucidate their mechanisms of action within the framework of the GCT. 4, 5 The growth of neurostimulation has been fueled in part by the increasing prevalence of neuropathic pain, 6 in particular the upsurge of patients with failed back surgery syndrome (FBSS), 7, 8 and the attempts to use strategies other than chronic opioid therapy to treat chronic neuropathic pain.Īlthough SCS technology has developed greatly in the past decades, 9 the last few years have witnessed the introduction of several novel devices and stimulation modalities, including high- frequency technology, 10, 11 dorsal root ganglion (DRG) stimulation, 12 burst stimulation, 13 and other paradigms. Today, an estimated 50,000 spinal cord neurostimulators are implanted annually. 1 The first reported clinical application of dorsal column stimulation came 2 years later, and the field has gradually expanded ever since. 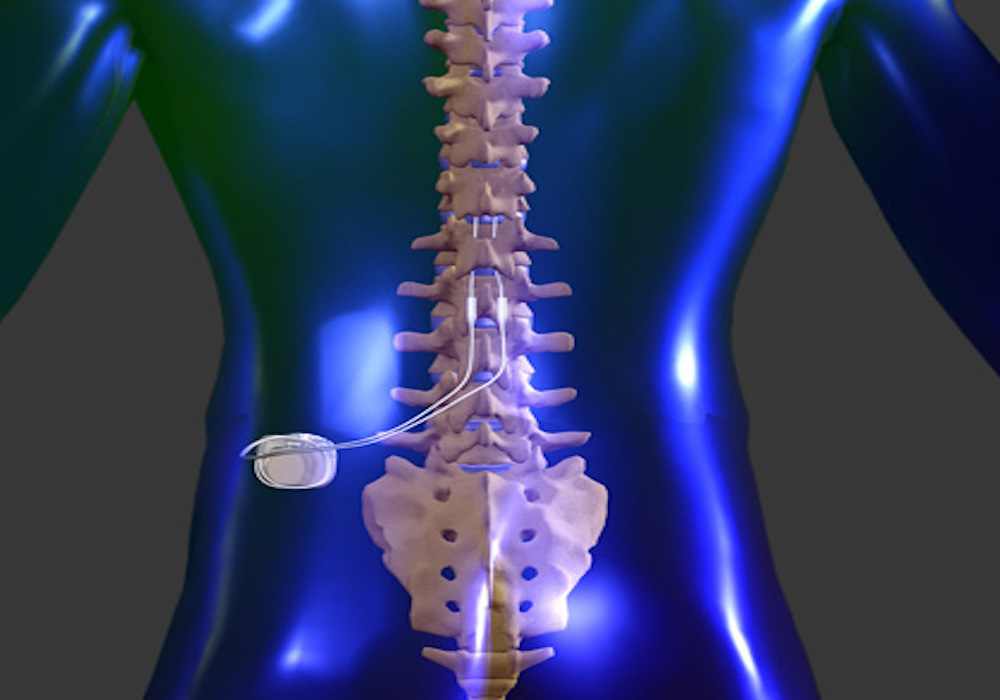
The field of spinal cord stimulation (SCS) owes its inception to the concept of gate control theory (GCT), put forth by Wall and Melzack in their landmark 1965 paper, which proposed that “control of pain may be achieved by selectively activating the large, rapidly conducting fibers”.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
